STEM CELL PROCEDURE
Get Free Online Consultation
How and Where Is My Therapy Done?
cells and exosomes
in modulating better orchestration as well as coordination of cellular events that are further responsible for 3R’s i.e. Repair, Regenerate and Rejuvenate. Accordingly, we have designed a protocol involving a cocktail of stem cells and their potential cargoes to reach the exact site of repair and promote healing.
Stem Cells Procedure: Advancells Protocol
The entire medical procedure at Advancells consists of 4 steps:
Source extraction
Isolation, analysis and concentration of the stem cells in the laboratory
Stem cell implantation
Post treatment care
Step 1- Qualification For The Therapy
Once you are counselled by our expert paneled doctor, and are convinced to go ahead with stem cell therapy; you will be prescribed some safety and efficacy assessments, which will further help us to know more about the severity of your disease. Further to which, some of the basic tests can also be prescribed that can help us to investigate, whether you are eligible for the treatment.
As per the regulatory guidelines proposed by CDESCO and GMP, you will be informed about the consequences, in the context of possible risks and benefits. Although, stem cell therapy has been referred to be as completely safe and risk-free procedure; under the moral and legal premise of informed consent, you have the right to make decisions about your own health and medical condition.
Pre-Treatment Assessments
- Routine Blood Tests
- Infectious Diseases Testing
- Urine Analysis
- Physical Examination
- X Ray
Pre-Operative Assessments
- Surgical Correction
- MRI
- CT Scan
- Medical History
Step 2- Source Extraction
As per our standardized protocol, discussed herewith specified amount of sample will be extracted from patient’s own body with the application of local anesthesia. The entire procedure should not take more than 7-8 hrs; however, in case of certain critical cases, patient may have to wait for a little longer period of time.
Bone Marrow Derived Adult Stem Cells Extraction
One of our senior Neurosurgeon/Orthopaedic surgeons will extract the bone marrow from the iliac crest (Hip Joint). Normally, the procedure takes around 30-45 minutes. Local anaesthesia is first administered on the area of the skin that is to be punctured. A bone marrow extraction needle is then used to extract about 100-120 ml of bone marrow. Option of general anaesthesia is available for patients who need it. Once the extraction is done, the sample will be immediately transferred to our clean room facility for processing.
More detailed information on the bone marrow collection procedure is available in the Bone Marrow Informed Consent document (PDF file).

Isolation of stem cells from Bone Marrow
In a clean room, stem cells will be separated from other unwanted mature blood cells and plasma. The processing of samples will be done as per cGMP standards under sterile conditions. Utmost care will be taken to make sure that stem cells do not get contaminated by any kind of infections. Post processing all samples will be tested for quality and quantity of stem cells.
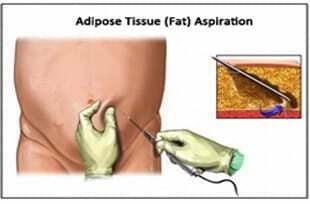
Adipose Tissue Derived Adult Stem Cells Extraction
One of our operating surgeon/cosmetic surgeons will extract an adipose tissue from an area of the body, which is generally pre-umbilical area; where sufficient amount of fat is available. Once the location is confirmed, a patient will be asked to lie in a supine position. He will be given a local anaesthesia. A 60 CC cannula is inserted under subcutaneous layer and approximately 100 CC adipose tissue is collected. Once the aspiration is complete, the sample will be immediately transferred to the clean room for further processing.
More detailed information on the adipose Tissue collection procedure is available in the Adipose Tissue Informed Consent document (PDF file)
Isolation of Stem Cells From Fat
Fat is abundant source of progenitor/stem cells; which will be separated from other mature cells such as macrophages, adipocytes etc. in our clean room facility. The processing of sample is done as per approved cGMP standards under sterile conditions. Utmost care is taken to make sure that stem cells do not get contaminated by any kind of infections. Post processing, all samples will be tested for quantity as well as quality.
Step 3- Laboratory Processing
All our laboratory procedures are automated and are performed in a completely closed system with a minimum manual interface. Each step is documented for analysis that can be retrieved at any point in time for further analysis.
As our client, you are valuable to us and we appreciate the trust you have shown in our abilities. As a token of that appreciation, we are happy to present all the evaluation reports to you on demand. Some of which are listed herewith.
If you are curious to know more about the procedure, kindly refer the detailed file:
Step 4- Stem Cell Implantation
Once stem cells are enriched and ready to be injected back into the body, we have worked on different modes of implanting them. However, depending upon the patient’s physiological and mental well being, one of the following will be finalized by our experts.
Our Established Quality Standards for Various Stem Cell Sources
In order to ensure the quality and effectiveness, we follow a safety evaluation system, based on the existing regulatory guidelines and advanced testing techniques. With the help of this system, we are comprehensively evaluating cell safety, quality in terms of viability, cell number and stemness markers, microbiological evaluation of the final release; not only in line with basic biological products, but also following general safety requirements of the patients undertaking cell-based therapies.
Stem Cell Implantation
Stem cells can be implanted in following major ways:
1. Intravenous Administration
Intravenous administration is the IV infusion of stem cells into the veins. IV and mannitol are infused in the blood of the patient through a drip. The primary purpose of Mannitol is to expand blood volume in the central nervous system; so that maximum cells will reach at the targeted area.
The entire IV Mannitol infusion session takes about 20 minutes. After this, the stem cells are slowly infused through that channel and they fuse with the blood and spread across the body. Before giving any IV medication, the following factors should be kept in mind:
- It is given to right patient
- Right dose is given
- Patient is not allergic to that dose
- It is given at right place and right time
More detailed information on the IV-Procedure is available in the IV-Mannitol Informed Consent Document (PDF File)
2. Intra-thecal Administration (Lumbar Puncture)
Intrathecal administration is the infusion of stem cells via injection into the spinal canal, most popularly known as lumbar puncture. By using this route of administration, blood brain barriers are avoided ensuring maximum deliver of cells into the cerebrospinal fluid. This route is particularly efficient in neurological disorders.
For the infusion of cells, puncture is made in the lumbar region; which is the lowest vertebrate at the back from where some spinal fluid is extracted. The administrator will ensure that the volume of the spinal fluid extracted will be equivalent to the amount of stem cells infused.
This will ensure that there is very little pressure difference in the spinal fluid space.
Any change in pressure of the fluids in the spinal cord may cause a little headache and dizziness in the patient. This symptom is not very hazardous and it lasts only for 1 or 2 days. However, many a times this pain can be uncomfortable especially while getting up from a horizontal position due to the change in the pressure ratios.
More detailed information on the LP procedure is available in the LP Informed Consent Document (PDF File)
Follow up Treatment
In a post treatment care, the patient will be asked to visit the doctors after a specific period of time, for some rehabilitation programmes including Behavioural and emotional development, Communication and social Interaction, Self regulation, Cognitive and Adaptive skills.
Treatment Disclaimer
Before undergoing this process; a patient must understand that stem cell therapy is an experimental therapy that may fail to work at certain times.
A patient might be refused treatment or the proposed protocol might change depending on the patient’s health condition at the time of procedure.
At Advancells, our primary motive is to offer the best technology available; safety of which has been proven across the world.
Treatable Diseases
Useful Links
Advancells
Partner With Us
Newsletter Subscription
“Advancells is a strong advocate of greater transparency in medical procedures in general and Stem Cells Procedures in particular. As a part of our effort of bringing transparent procedures to our clients, we provide each client a Third Party Certificate (from an internationally accredited lab) for the cell count and viability of the cells that we are able to process from the autologous cell source of the patient. To view the sample copy of the certificate “click here“
By Advancells. Copyright 2021. All Rights Reserved. All images and content used in this site are original. Any attempt to copy will come under criminal jurisdiction.



