
Stem Cells Treatment Procedure
- Pre-treatment counselling on the stem cell therapy.
- Formulate treatment plan.
- Stem cell extraction and administration.
- Post-treatment Care.
How and Where Is My Therapy Done?
Making a decision to undergo stem cell therapy is a huge step. Advancells truly admires your spirit of being healthier you!
Get Free Consultation
We offer a personalized treatment plan, depending upon individual marker assessments and other clinical evaluations. Cellular science has demonstrated active involvement of stem cells and exosomes in modulating better orchestration as well as coordination of cellular events that are further responsible for 3R’s i.e. Repair, Regenerate and Rejuvenate. Accordingly, we have designed a protocol involving a cocktail of stem cells and their potential cargoes to reach the exact site of repair and promote healing.
Stem Cell Treatment Procedure: Advancells Protocol
The entire medical procedure at Advancells consists of 4 steps:



Step 1: Qualification For The Therapy
Once you are counselled by our expert paneled doctor, and are convinced to go ahead with stem cell therapy; you will be prescribed some safety and efficacy assessments, which will further help us to know more about the severity of your disease. Further to which, some of the basic tests can also be prescribed that can help us to investigate, whether you are eligible for the treatment.
As per the regulatory guidelines proposed by CDESCO and GMP, you will be informed about the consequences, in the context of possible risks and benefits. Although, stem cell therapy has been referred to be as completely safe and risk-free procedure; under the moral and legal premise of informed consent, you have the right to make decisions about your own health and medical condition.
Pre-Treatment Assessments
- Routine Blood Tests
- Infectious Diseases Testing
- Urine Analysis
- Physical Examination
- X Ray

Pre-Operative Assessments
- Surgical Correction
- MRI
- CT Scan
- Medical History
Step 2: Source Extraction
As per our standardized protocol, discussed herewith specified amount of sample will be extracted from patient’s own body with the application of local anesthesia. The entire procedure should not take more than 7-8 hrs; however, in case of certain critical cases, patient may have to wait for a little longer period of time.
Bone Marrow Derived Adult Stem Cells Extraction
One of our senior Neurosurgeon/Orthopaedic surgeons will extract the bone marrow from the iliac crest (Hip Joint). Normally, the procedure takes around 30-45 minutes. Local anaesthesia is first administered on the area of the skin that is to be punctured. A bone marrow extraction needle is then used to extract about 100-120 ml of bone marrow. Option of general anaesthesia is available for patients who need it. Once the extraction is done, the sample will be immediately transferred to our clean room facility for processing.
More detailed information on the bone marrow collection procedure is available in the Bone Marrow Informed Consent document (PDF file).
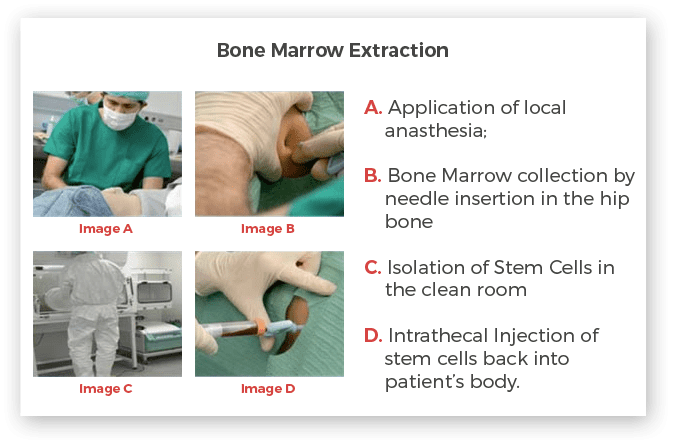
Isolation of stem cells from Bone Marrow
In a clean room, stem cells will be separated from other unwanted mature blood cells and plasma. The processing of samples will be done as per cGMP standards under sterile conditions. Utmost care will be taken to make sure that stem cells do not get contaminated by any kind of infections. Post processing all samples will be tested for quality and quantity of stem cells.
Adipose Tissue Derived Adult Stem Cells Extraction
One of our operating surgeon/cosmetic surgeons will extract an adipose tissue from an area of the body, which is generally pre-umbilical area; where sufficient amount of fat is available. Once the location is confirmed, a patient will be asked to lie in a supine position. He will be given a local anaesthesia. A 60 CC cannula is inserted under subcutaneous layer and approximately 100 CC adipose tissue is collected. Once the aspiration is complete, the sample will be immediately transferred to the clean room for further processing.
More detailed information on the adipose Tissue collection procedure is available in the Adipose Tissue Informed Consent document (PDF file)
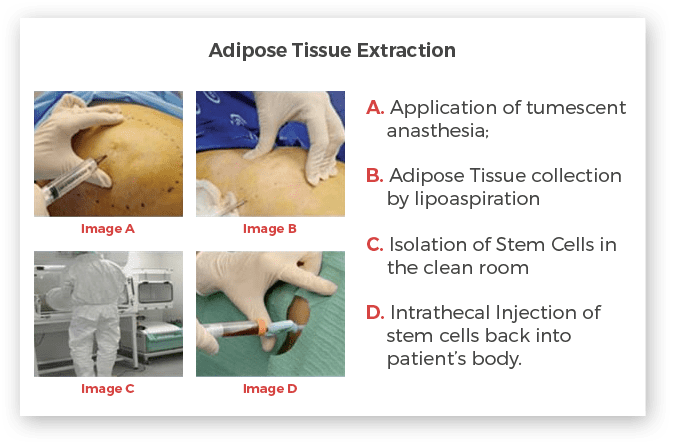
Isolation of Stem Cells From Fat
Fat is abundant source of progenitor/stem cells; which will be separated from other mature cells such as macrophages, adipocytes etc. in our clean room facility. The processing of sample is done as per approved cGMP standards under sterile conditions. Utmost care is taken to make sure that stem cells do not get contaminated by any kind of infections. Post processing, all samples will be tested for quantity as well as quality.
Step 3: Laboratory Processing
All our laboratory procedures are automated and are performed in a completely closed system with a minimum manual interface. Each step is documented for analysis that can be retrieved at any point in time for further analysis.
As our client, you are valuable to us and we appreciate the trust you have shown in our abilities. As a token of that appreciation, we are happy to present all the evaluation reports to you on demand. Some of which are listed herewith.
If you are curious to know more about the procedure, kindly refer the detailed file:
Step 4: Stem Cell Implantation
Once stem cells are enriched and ready to be injected back into the body, we have worked on different modes of implanting them. However, depending upon the patient’s physiological and mental well being, one of the following will be finalized by our experts.
Our Established Quality Standards for Various Stem Cell Sources
In order to ensure the quality and effectiveness, we follow a safety evaluation system, based on the existing regulatory guidelines and advanced testing techniques. With the help of this system, we are comprehensively evaluating cell safety, quality in terms of viability, cell number and stemness markers, microbiological evaluation of the final release; not only in line with basic biological products, but also following general safety requirements of the patients undertaking cell-based therapies.
Stem Cell Implantation
For optimum therapeutic performance, stem cells can be administered in the patients’ body via different routes:
1. Intravenous Administration
Intravenous (IV) administration is the infusion of stem cells via the patient’s veins. Mannitol is used as a supplementary agent with IV drip to expand the blood volume around central nervous system. This ensures optimum efficacy with maximum no. of cells reaching the target area. Intravenous administration image.
Intravenous (IV) administration is the infusion of stem cells via the patient’s veins. Mannitol is used as a supplementary agent with IV drip to expand the blood volume around central nervous system. This ensures optimum efficacy with maximum no. of cells reaching the target area:
- Right dose is given to the right patient
- Patient is not allergic to IV dose
- The infusion is done aseptically at the right time
More detailed information on the IV administration procedure is available in the IV-Mannitol Informed Consent Document (PDF File)
2. Intrathecal Administration (Lumbar Puncture)
Intrathecal administration involves stem cell infusion via spinal canal injections, popularly known as lumbar puncture. Using this route of administration, optimum delivery of stem cells to the cerebrospinal fluid can be achieved without being affected by the blood-brain barrier. This route is particularly efficient in case of neurological disorders.
For the infusion process, a small volume of spinal fluid is extracted from the lumbar region and an equivalent volume of stem cells is implanted in the same vertebra region. This nullifies the pressure difference in the spinal fluid column.
Patients may experience mild side effects of dizziness and headache to cope up with the spinal fluid pressure difference. These symptoms may last only for 1-2 days and can be slightly prominent while getting up from a horizontal sleeping position. Rest assured, these symptoms do not pose any risk to the patients’ health.
More detailed information is available in the LP Informed Consent Document (PDF File)
3. Intra-Muscular Administration
Intra muscular injection is infusion of stem cells directly into the muscles. It is used in particular when small amount of cells are to be administered. The procedure is through injection ensuring release of pool of stem cells directly to the affected area. Target area with ailing tissues can include knee, shoulders, hips etc. Hence the ideal site for administering the injection are Thighs, Top of upper arm, Knee, Hips.
Once the targeted muscular injection is administered, the pool of stem cells reach the diseased area and effectively begin the process of rejuvenation and repair. Clinical results have further indicated that a combination of intramuscular injections with more than one infusion protocol is much more beneficial in promoting the regeneration and functional recovery of musculoskeletal environment.
4. Intra-Arterial Administration via catheter

Intra-arterial infusion of stem cells is performed through a thin catheter inserted into the patient’s artery. Prior to infusion, the catheter is very carefully directed towards the targeted area to ensure maximum cell delivery for optimum efficiency. This route of administration is generally preferred for vascular organs such as kidney, heart, and pancreas.
5. Intravitreal Infusion

The intravitreal drug delivery has become the most reformed technology in treating some of the ophthalmic conditions such as macular oedema, retinal vein occlusions, etc. These are the conditions wherein single dose administered through intravitreal region is more effective than repeated injections through retrobulbar mode.
The inside of the human eye is filled with a jelly like fluid known as “Vitreous humor ”. An intravitreal injection is a direct shot of stem cells into the vitreous structure near the retina, at the back of the eye. The entire procedure takes no more than 15-20 minutes.
- The procedure requires dilation of the pupil by placing specific eye drops in the eyes.
- Once pupils are dilated, the patient will be allowed to lie in a comfortable position and anaesthetic drops will be provided.
- After cleaning both the eyes and the eyelids using the iodine antiseptic solution, the eyes are kept open during the entire procedure with the help of a small device.
- A shot of stem cells will be injected directly into the vitreous region with the help of small needle; a patient may feel slight pressure on the eye without any pain.
- The eyes will be checked properly and some antibiotic ointment might be placed in the eyes, if required.
6. Retrobular Infusion of Cells

The retrobulbar injection procedure is routinely exercised by many ophthalmologists for the infusion of stem cells to treat eye diseases such as, Macular Degeneration, Glaucoma, and other disorders.
In this procedure, a specific needle is carefully inserted inferior to the maximum eye diameter perpendicular to your face, under local anesthesia conditions. Once past the axis, the needle is angled medially and superiorly before finally positioning within the muscle cone. Following that, a pool of viable stem cells are injected near the macula region, under the eye.
This method of stem cell administration is preferred for eye disorders as a higher number of cells can be directly targeted to optimize the output. In some cases, a combination of two protocols may be suggested by the experts in the best interest of patient well-being.
7. Liberation Angioplasty for Multiple Sclerosis CCSVI
 CCSVI Treatment Step 1 – Liberation Angioplasty
CCSVI Treatment Step 1 – Liberation Angioplasty
Liberation Angioplasty is a very critical process. X-ray scanning is used to guide the catheter with the balloon tip to the affected area or veins. Once it reaches the targeted area, the tip expands resulting in the widening of the stenotic area. The entire process takes around 90 minutes. Then the patients are kept in recovery room for around 4 hours to ensure that there is no bleeding from the catheter insertion tract.
CCSVI Treatment Step 2 – Insertion of Stem Cells
After the first step of liberation angioplasty the blood flows with ease through the stenotic veins. This is essential in order to determine the effective potential of the stem cells introduced to transform into specialized cells necessary for regeneration or repair of damaged tissue of the brain or the spinal cord. This procedure can also reduce the inflammation throughout the body. Many of the patients have reported that the recovery is extremely fast after this treatment.
8. Intra-dermal Administration
 Around the skin wounds :- Intra-dermal administration for wounds is specifically used in case of open sores such as diabetic foot or pressure ulcers. The procedure involves direct infusion of stem cells into or around the wound area, e.g., dermal layer of the skin. The skin dermal layer is the most preferred layer as it is highly vascularized and contains a high concentration of immune cells. Through this route, infused stem cells can promptly elicit and strengthen the body’s natural healing signals for faster recovery.
Around the skin wounds :- Intra-dermal administration for wounds is specifically used in case of open sores such as diabetic foot or pressure ulcers. The procedure involves direct infusion of stem cells into or around the wound area, e.g., dermal layer of the skin. The skin dermal layer is the most preferred layer as it is highly vascularized and contains a high concentration of immune cells. Through this route, infused stem cells can promptly elicit and strengthen the body’s natural healing signals for faster recovery.
 Around the hair follicles :- In this procedure, Platelet Rich Plasma (PRP) along with a pool of stem cells is infused around the hair follicles using micro-needles, after local anesthesia of the scalp area. This intra-dermal, local application of PRP with stem cells can accelerate the healing process around the follicles immediately to stimulate the native stem cells. These native cells, in turn, provide strength, support, vitality and resilience to the follicles and help in generating natural new hair strands.
Around the hair follicles :- In this procedure, Platelet Rich Plasma (PRP) along with a pool of stem cells is infused around the hair follicles using micro-needles, after local anesthesia of the scalp area. This intra-dermal, local application of PRP with stem cells can accelerate the healing process around the follicles immediately to stimulate the native stem cells. These native cells, in turn, provide strength, support, vitality and resilience to the follicles and help in generating natural new hair strands.
 Platelet Rich Plasma for facial skin :- In this procedure, Platelet Rich Plasma (PRP) along with a pool of stem cells is infused using micro-needles, after local numbing of the face area. The face is locally numbed using numbing cream and the infusion is accordingly administered at different facial regions such as around the eyes, lips, chin, etc. This intra-dermal, local application of PRP with stem cells can accelerate the healing process immediately to stimulate the native stem cells. These native cells enhance the production of collagen and the removal of photo-damaged skin cells, pigmentations, and wrinkles, giving way to a radiant natural skin.
Platelet Rich Plasma for facial skin :- In this procedure, Platelet Rich Plasma (PRP) along with a pool of stem cells is infused using micro-needles, after local numbing of the face area. The face is locally numbed using numbing cream and the infusion is accordingly administered at different facial regions such as around the eyes, lips, chin, etc. This intra-dermal, local application of PRP with stem cells can accelerate the healing process immediately to stimulate the native stem cells. These native cells enhance the production of collagen and the removal of photo-damaged skin cells, pigmentations, and wrinkles, giving way to a radiant natural skin.
In general, most of the intradermal injections are delivered inside the skin through micro-needles using mantoux technique (the needle is inserted at an angle of 5-15 degree).
9. Tissue Reconstruction through Biodegradable Scaffolds
 Through the advanced developments in the field of Tissue Engineering, regeneration of damaged tissue is possible by allowing rapid cellular integration into an optimized biocompatible scaffold.
Through the advanced developments in the field of Tissue Engineering, regeneration of damaged tissue is possible by allowing rapid cellular integration into an optimized biocompatible scaffold.
These porous three-dimensional scaffolds essentially act as tissue regeneration templates where stem cells are implanted with PRP. Thus, the regeneration process is systematically allowed through a triad combination of stem cells, signaling pathways and biodegradable material.
Different Types of Scaffolds that can be used in tissue reconstruction
Autograft or Autologous Bone Graft is constructed using the patient’s own bone tissue. An autograft tissue is typically obtained from the chin, jaw, lower leg bone, hip, and skull. The main advantage of an autograft is the presence of living cellular elements that enhance bone growth. However, one of the major drawbacks of autografts is the requirement of a second procedure for bone harvest.
Allograft or Allogenic Bone Graft is basically a dead bone harvested from a cadaver, which is further processed to form a framework or scaffold to support bone regeneration from the surrounding tissue. The main advantage of an allograft is its ability to support stem cell culture in the form of a good biocompatible scaffold
Xenograft of Xenogenic Bone Graft is derived from the living bone of other species, such as cow or pig. This type of bone graft is processed at a very high temperature to avoid any potential immune rejection or contamination. Similar to allografts, xenografts can also be used as bio-scaffolds.
10. Surgical Administration for patients having stroke or SCI
Brain-Surgery
Brain surgery for stem cell infusion requires a lot of modern technical equipment and tools in order to operate and infiltrate the skull (Craniotomy). A highly valued advanced brain navigation system is used in order to reduce any kind of brain tissue damage during surgery. Strict protocols of anesthesia and surgery are followed for this procedure of stem cell administration. After the surgery, patients are generally kept under observation in the care unit for a night and thereafter shifted to the normal ward, prior to being discharged from the hospital.
Surgery of the Spine
Spinal surgeries for stem cell infusion are also very critical and require ultramodern technical equipment to operate and infiltrate the spinal cord/vertebrae (Laminectomy). In this procedure, after the spinal column is incised, a further incision is done on the embedded dura-mater. Following that, a pool of viable stem cells is infused using a very fine needle and the incisions are sealed accordingly. Strict protocols of anesthesia and surgery are followed for this procedure of stem cell administration. After the surgery, patients are generally kept under observation in the care unit for a night and thereafter shifted to the normal ward, prior to being discharged from the hospital.
Postoperative Care
Patients can leave the hospital after few hours of stem cell transplantation although in certain cases especially when cells are transplanted Intra-arterial via catheter or surgeries; a patient is advised to stay for a longer duration.
A 24-hour hotline is available for the patients to clear up any doubts and queries that they may face right after their discharge. Our doctors and patient consultants are always available over phone and emails. A patient can easily be in touch with his or her physician, which in return helps us to study their recovery.
Treatment Disclaimer
Before undergoing this process; a patient must understand that stem cell therapy is an experimental therapy that may fail to work at certain times.
A patient might be refused treatment or the proposed protocol might change depending on the patient’s health condition at the time of procedure.
At Advancells, our primary motive is to offer the best technology available; safety of which has been proven across the world.
Advancells is a strong advocate of greater transparency in medical procedures in general and Stem Cells Procedures in particular. We do not have any medical doctors on our payroll and we do not give any medical opinion or conduct medical procedures at our premises. We are a CGMP compliant cell manufacturing facility and provide each client a Third Party Certificate (from an internationally accredited lab) for the cell count and viability of our cells. View sample Certificate


